Advancing IPF Treatment with a Novel Autotaxin Inhibitor and AI-Driven Precision Analysis
Bridge Biotherapeutics, which secured a landmark billion deal in 2019, faced a setback in October 2020 when its partnership with Boehringer Ingelheim for idiopathic pulmonary fibrosis (IPF) drug candidate 무료 슬롯 머신 다운 받기877 was terminated. Four years later, the company is regaining momentum, earning a main stage presentation at the JPM Healthcare Conference, a sign of renewed confidence in the drug’s potential.
Currently in Phase 2 global trials, Bridge Biotherapeutics plans to disclose top-line data soon. CEO Jung-kyu Lee, in a recent interview with Hit News, confirmed ongoing discussions regarding a global licensing agreement and emphasized the company’s strengthened belief in 무료 슬롯 머신 다운 받기877’s efficacy. Since regaining full rights, the company has independently refined its clinical strategy, focusing on long-term efficacy through extended dosing regimens.

무료 슬롯 머신 다운 받기877: A First-in-Class Autotaxin Inhibitor Targeting IPF
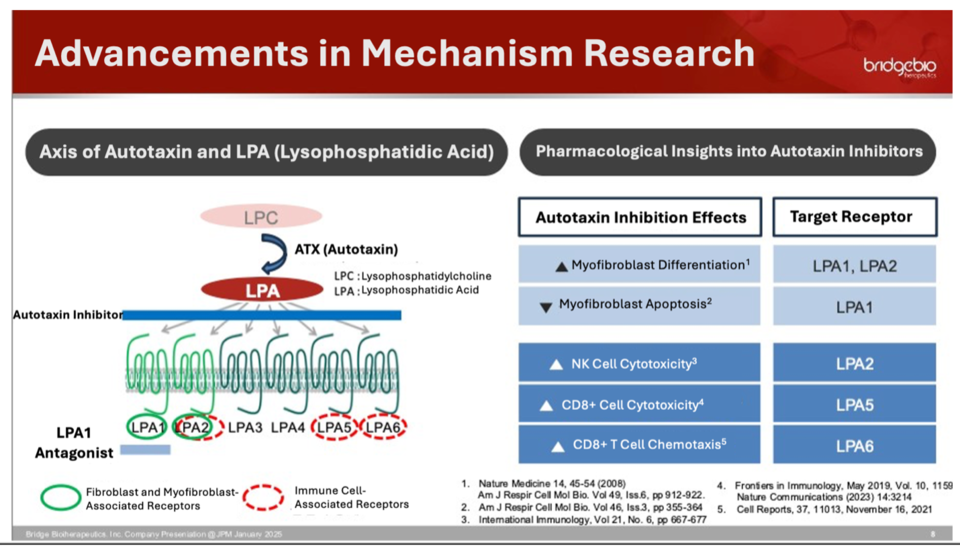
무료 슬롯 머신 다운 받기877 selectively inhibits autotaxin, a plasma protein linked to inflammation and fibrosis. CEO Lee highlighted three key factors behind the drug’s development: the novelty of autotaxin inhibition, minimal competition in the field, and the high unmet medical need in IPF, a severe and rare disease.
Current IPF treatments, Ofev (nintedanib) and Esbriet (pirfenidone), slow disease progression but do not offer a cure and often cause significant side effects. Unlike these therapies, 무료 슬롯 머신 다운 받기877 is designed not only to suppress myofibroblast formation but also to clear accumulated myofibroblasts, potentially modifying the disease course.
By inhibiting autotaxin, 무료 슬롯 머신 다운 받기877 reduces lysophosphatidic acid (LPA) levels, which play a critical role in fibrosis progression. This dual mechanism positions 무료 슬롯 머신 다운 받기877 as a promising candidate capable of both halting disease advancement and potentially reversing fibrosis.
“If our clinical trials validate this advantage, 무료 슬롯 머신 다운 받기877 could establish itself as a highly competitive and transformative therapy in the IPF market,” Lee stated.
무료 슬롯 머신 다운 받기877’s Strength? Efficacy-Validating Biomarker
A key advantage of 무료 슬롯 머신 다운 받기877 is its biomarker-based validation approach. Lee explained, “Biomarkers provide a measurable indicator of a drug’s action and effectiveness.” 무료 슬롯 머신 다운 받기877’s efficacy can be assessed by tracking LPA levels in the bloodstream, offering real-time insights into treatment response.
Unlike existing IPF drugs evaluated over 12-week trials, 무료 슬롯 머신 다운 받기877’s 24-week study provides a more comprehensive analysis of long-term effects. Given that IPF treatment typically requires extended therapy, these findings will serve as a critical benchmark for real-world therapeutic potential.
Enhancing Clinical Precision with AI-Based Imaging Analysis
무료 슬롯 머신 다운 받기 Biotherapeutics has integrated Brainomix’s AI-based e-ILD technology into its Phase 2 trials to improve clinical precision. This tool analyzes high-resolution computed tomography (HRCT) images, enabling a systematic and quantitative evaluation of disease progression.
Traditional IPF trials rely on forced vital capacity (FVC) measurements, which can be inconsistent due to patient variability. AI-driven imaging analysis provides a more objective assessment of lung fibrosis, complementing FVC measurements for enhanced accuracy.
Lee noted, “By incorporating AI-based analysis, we ensure a more reliable evaluation of 무료 슬롯 머신 다운 받기877’s efficacy.” While detailed efficacy data remains blinded, he expressed confidence that positive results will drive strong market interest and potential out-licensing agreements for Phase 3 trials.
Return Brought Expertise: Advancing 무료 슬롯 머신 다운 받기301 & Pipeline
Despite the challenges following Boehringer Ingelheim’s return of 무료 슬롯 머신 다운 받기877, Bridge Biotherapeutics leveraged the experience to refine its clinical expertise. “Collaborating with a global leader in IPF provided invaluable insights, reinforcing our confidence in 무료 슬롯 머신 다운 받기877,” Lee stated. Independent research reaffirmed the promise of autotaxin inhibition, guiding strategic refinements.
The company operates the Boston Discovery Center (BDC), a global R&D hub, and collaborates with over 50 clinical sites across five countries for its Phase 2 trials. Post-licensing, 무료 슬롯 머신 다운 받기 Biotherapeutics plans to expand BDC operations and establish a fully integrated drug discovery platform.
In parallel, the company is advancing 무료 슬롯 머신 다운 받기301, an IPF candidate that increases cAMP levels. Developed through drug repositioning, 무료 슬롯 머신 다운 받기301 repurposes an existing CNS drug for IPF, accelerating its clinical pathway.
Following a Pre-IND meeting, the company secured a Phase 1 trial waiver and plans to submit a Phase 2 IND application to the FDA this year.
Lee emphasized, “Our decade-long expertise in clinical trial design and execution with 무료 슬롯 머신 다운 받기877 will serve as a foundation for advancing next-generation fibrosis treatments and strengthening global competitiveness.”
